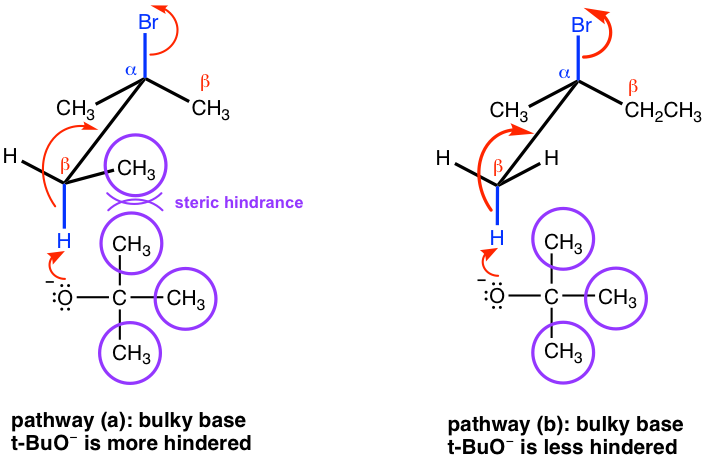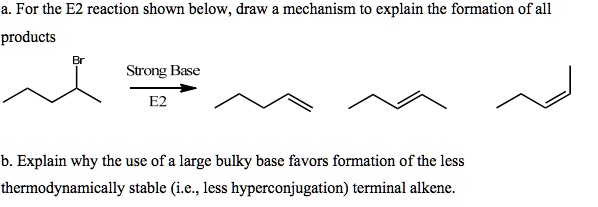
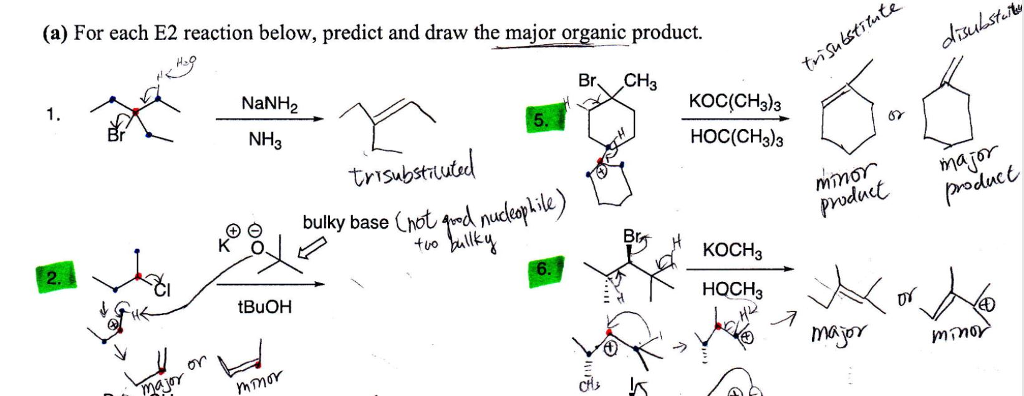
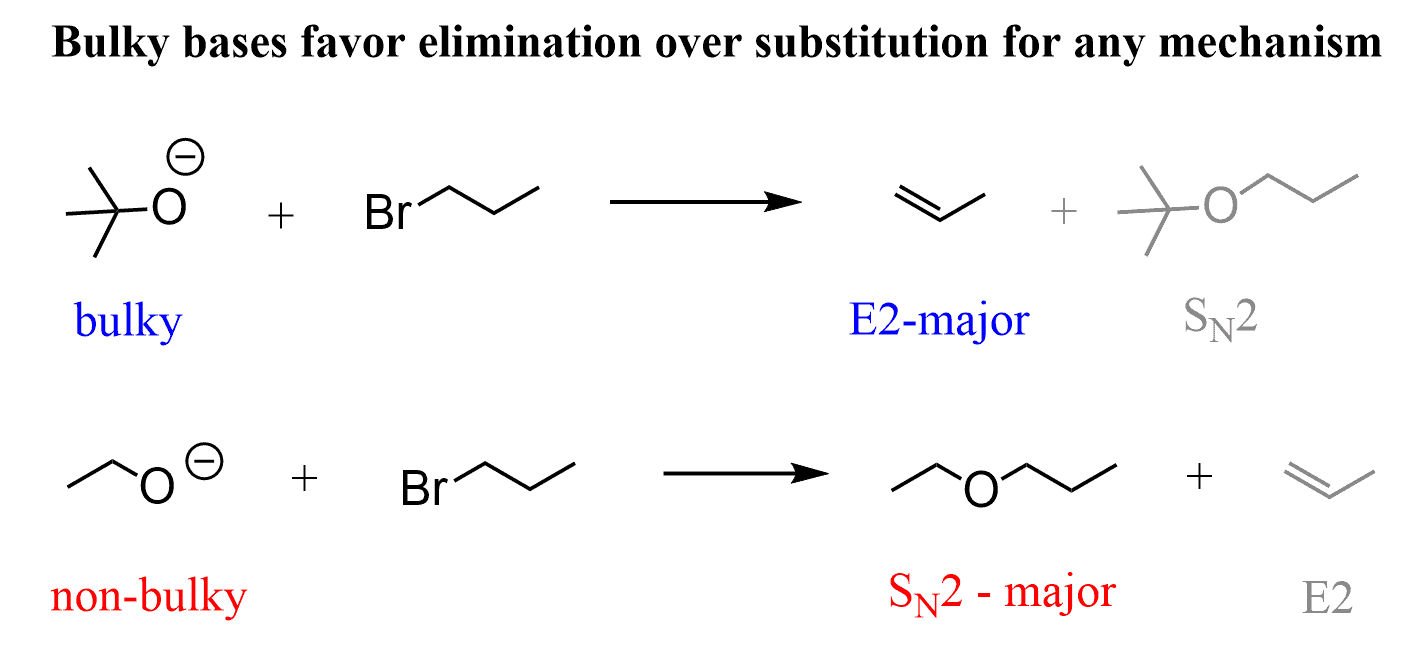
SOLVED: For thc E2 rcaction shown bclow, draw mechanism to cxplain the formation of all products Strong Base Explain why the usc of a largc bulky base favors formation of the less
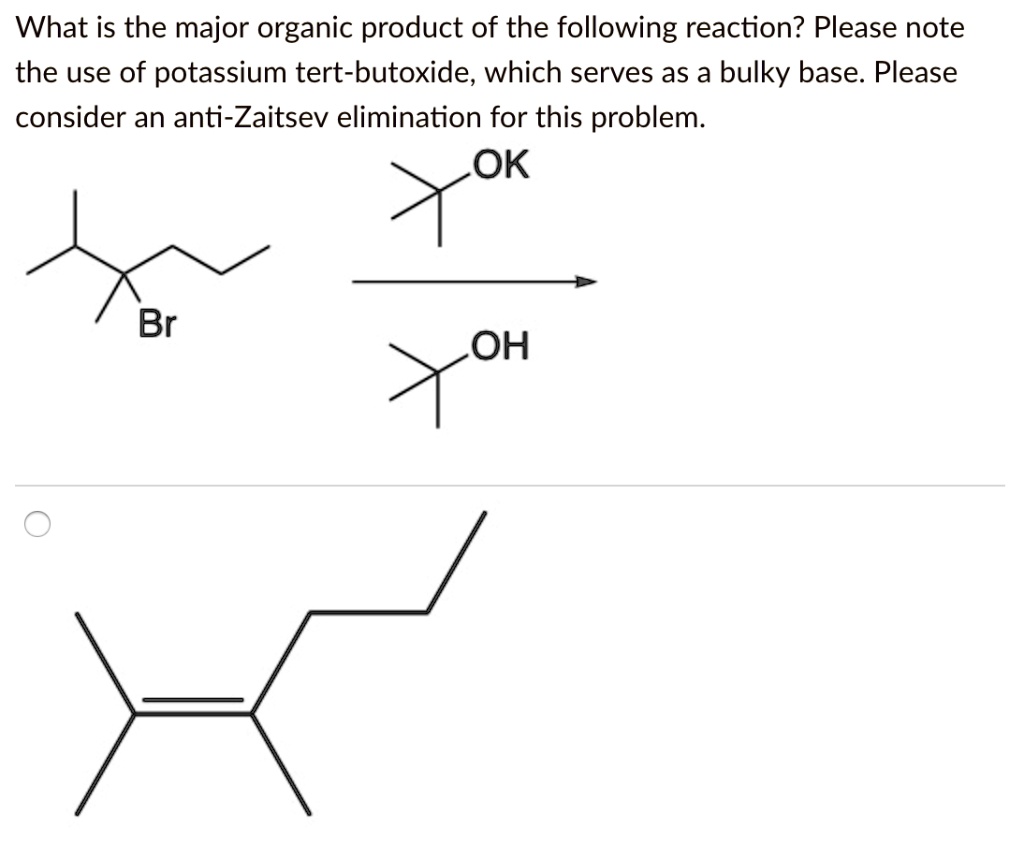
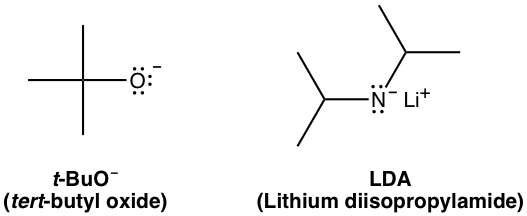
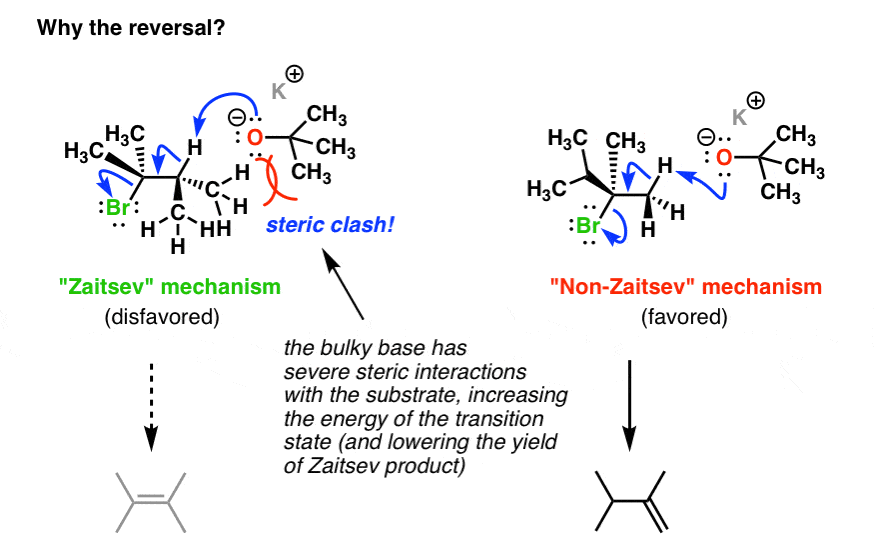
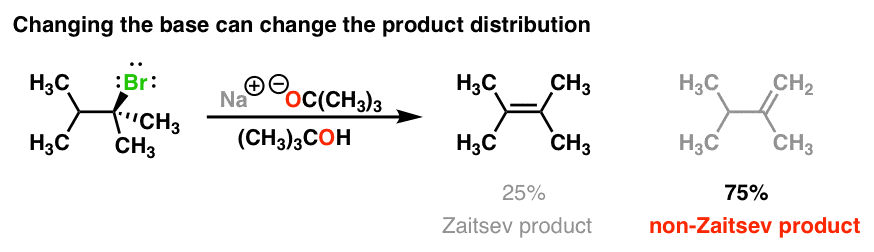
SOLVED: What is the major organic product of the following reaction? Please note the use of potassium tert-butoxide; which serves as a bulky base. Please consider an anti-Zaitsev elimination for this problem:
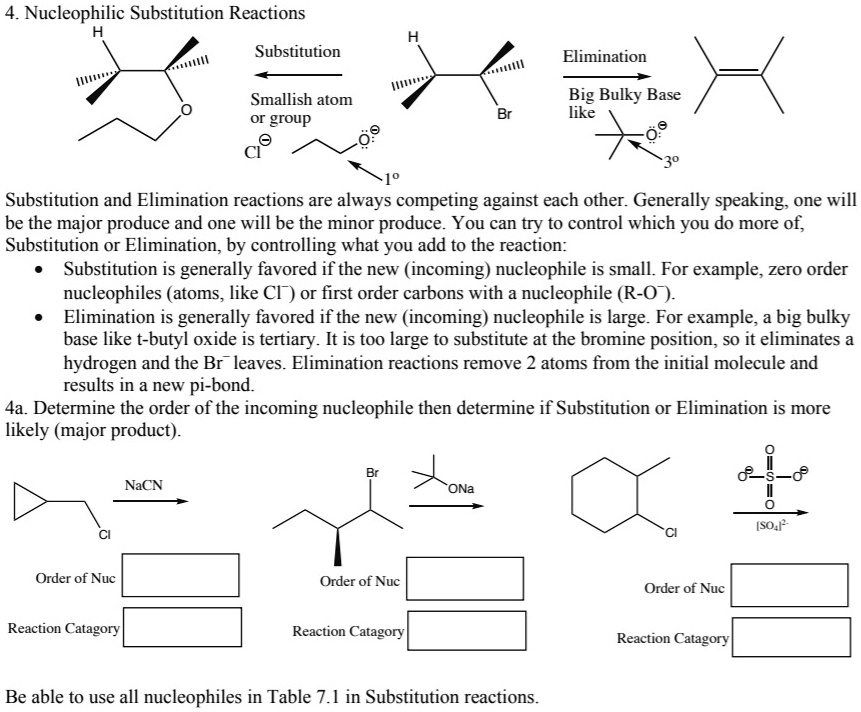
SOLVED: 4. Nucleophilic Substitution Reactions Substitution Elimination Smallish atom or group Big Bulky Base like Substitution and Elimination reactions are always competing against each other. Generally speaking; one will be the major
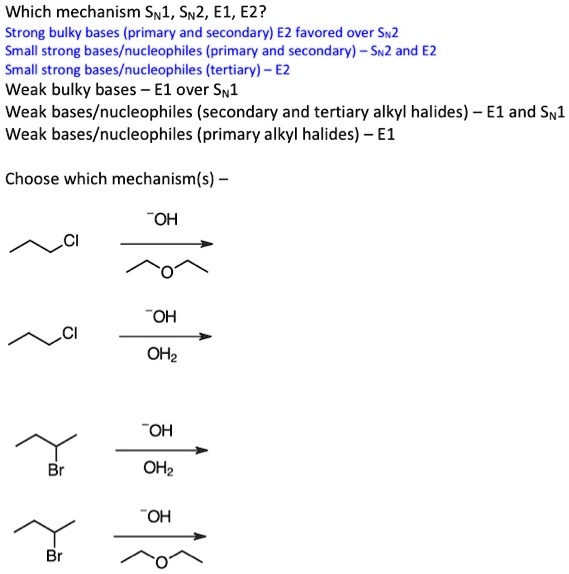
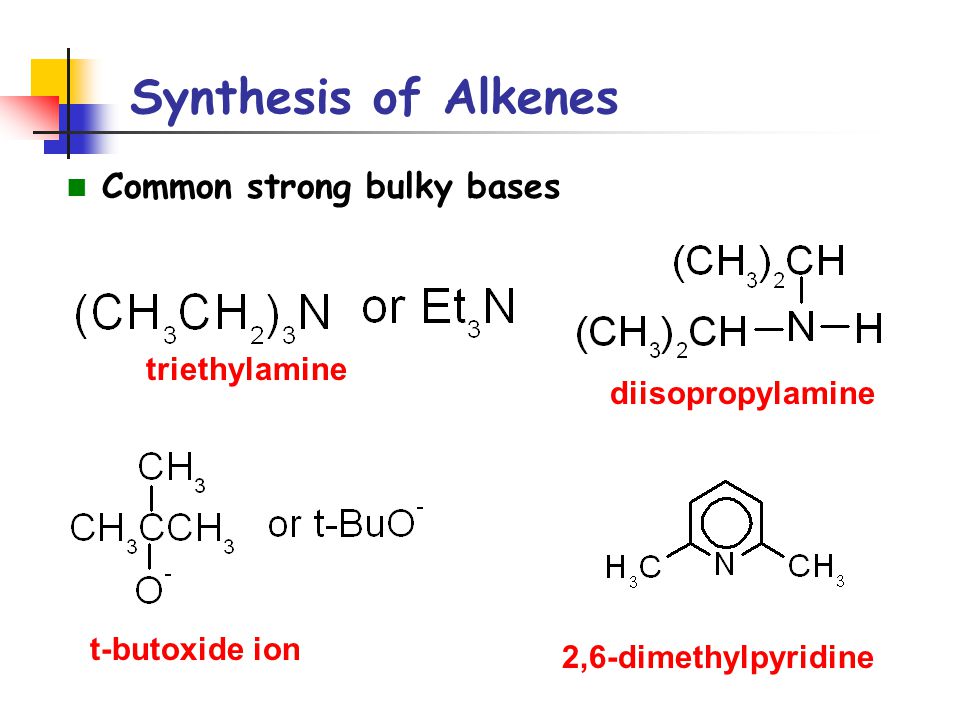
SOLVED: Which mechanism Swl, Sn2, E1, E2? Strong bulky bases (primary and secondary) E2 favored over Sx2 Small strong bases/nucleophiles (primary and secondary) Sn2 and E2 Small strong bases/nucleophiles (tertiary) Weak bulky







:max_bytes(150000):strip_icc()/most-common-strong-bases-603649-ADD-Final2-a2c0ac3120ff4b65bd98989ee298878c.png)